Discover out what clicks on FoxBusiness.com.
A well-liked model of eye drops is recalled throughout the country as a result of potential contamination, which may trigger visible injury, in line with the Meals and Drug Administration (FDA).
On Monday, the FDA introduced that Texas-based Alcon Laboratories was voluntarily recalling a single lot of “Systane Lubricant Eye Drops Extremely SPF, Single Vials On-the-Go” as a result of the merchandise could also be contaminated with fungus.
The corporate reported a client criticism {that a} “international object” was discovered inside a sealed, single-use bottle and decided the fabric was “fungal in nature.”
Fungal contamination of eye merchandise is thoughto potentially cause eye infections, the FDA mentioned.
EYE PRODUCTS SOLD AT WALMART, CVS MAY POSE AN INFECTION RISK

The Meals and Drug Administration (FDA) introduced Monday that Texas-based Alcon Laboratories is voluntarily recalling a single lot of “Systane Lubricant Eye Drops Extremely SPF, Single Vials On-the-Go” as a result of the merchandise could also be contaminated with enjoyable substances. (The Meals and Drug Administration (FDA) / Fox Information)
If an an infection happens, the FDA mentioned it may threaten imaginative and prescient and, in very uncommon instances, probably be life-threatening in immunocompromised sufferers.
To this point, the FDA mentioned Alcon Laboratories has not obtained any stories of adversarial reactions associated to this recall.
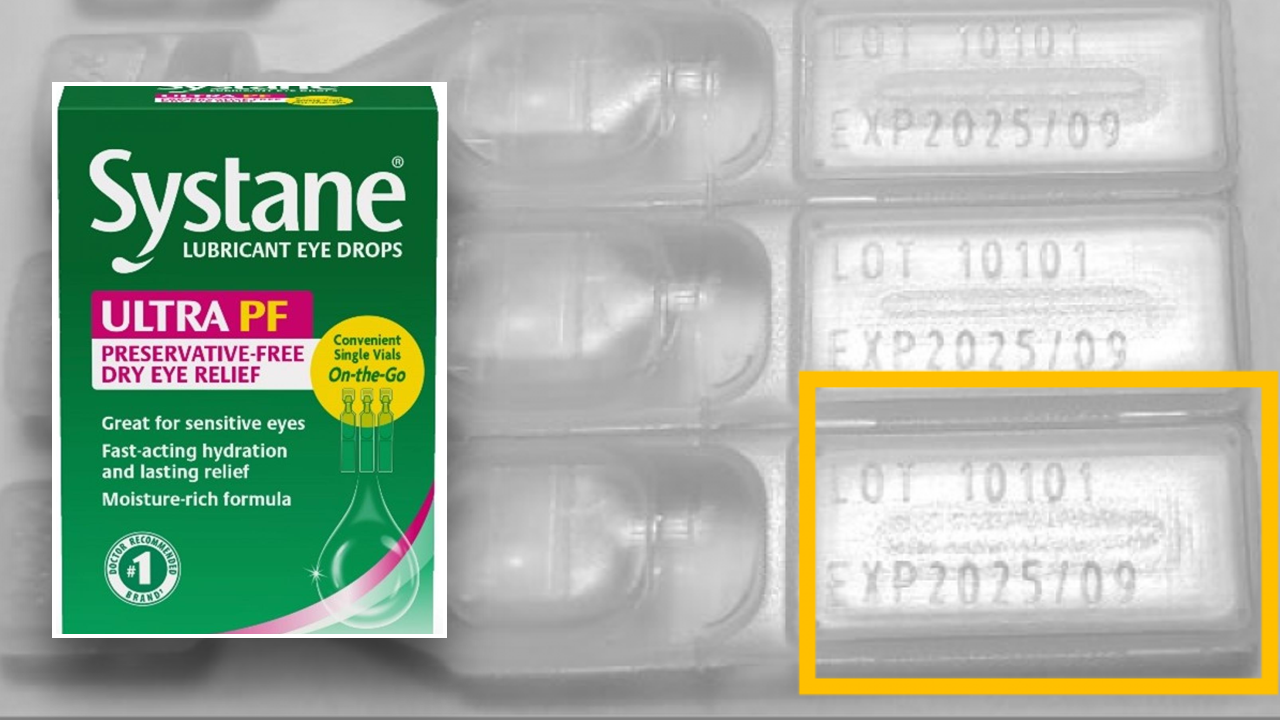
The FDA says the affected product consists of Systane Lubricant Eye Drops Extremely PF, Single Take-Away Bottles, 25 items and is proscribed to lot quantity 10101, expiration date 2025/09.
The product may be recognized by the inexperienced and pink carton design, the presence of the model names “Systane” and “ULTRA PF” on the entrance of the carton, and the bundle dimension “25 vials,” the FDA defined in a press launch.
FDA RAISES COSTCO EGG RECALL TO HIGHEST RISK LEVEL DUE TO SALMONELLA FEARS

“Systane Extremely SPF Lubricating Eye Drops, Single On-The-Go Bottles” have been recalled as a result of the merchandise could also be contaminated with fungus, in line with the FDA. (The Meals and Drug Administration (FDA) / Fox Information)
A lot of the probably affected eye drops had been additionally distributed nationwide to stores and over the Web.
Shoppers who possess the recalled eye drops are urged to cease utilizing them instantly and return them to the place of buy for a substitute or refund, the FDA mentioned.
Distributors or retailers who’ve the recalled eye drops are additionally requested to eliminate any remaining inventory of the contaminated product.
Alcon Laboratories additionally notifies its distributors and prospects by letter, electronic mail and/or phone name and arranges for substitute of all recalled merchandise.
BELOVED SNACK BRAND RECALLS ‘LIMITED’ NUMBER OF POPULAR CHIPS BAGS DUE TO UNDECLARED ALLERGEN: FDA

Alcon Laboratories issued the recall after a client complained of a “international object” present in a sealed bottle of eye drops. (The Meals and Drug Administration (FDA) / Fox Information)
This newest recall comes as a number of eye merchandise have been pulled from cabinets over the previous yr because of the potential threat of an infection.
In February, eye ointments bought at CVS and Walmart stores nationwide had been recalled after the FDA found a “lack of sterility assurance” on the manufacturing plant.

Alcon Laboratories in Texas is voluntarily recalling Systane Extremely PF Lubricating Eye Drops following a client criticism concerning international materials that could be noticed inside a sealed bottle. (The Meals and Drug Administration (FDA) / Fox Information Latino)
The 4 affected merchandise, that are meant to be sterile, are bought underneath the manufacturers Equate, CVS Well being and AACE Prescription drugs, and have expiration dates starting from February 2024 to September 2025. The merchandise had been distributed nationwide to the wholesalers, to retailers and thru the product distributor, Walmart, CVS and AACE Pharmaceuticals Inc.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
In November 2023, Kilitch Healthcare India Restricted voluntarily recalled 27 eye drops, all of which had been on the FDA’s rolling record of merchandise that could be contaminated with micro organism, posing a threat of eye an infection and imaginative and prescient loss.
Not one of the remembers talked about above are linked to the outbreak of antibiotic-resistant micro organism. pseudomonas aeruginosa associated to eye merchandise by World Pharma Healthcare.
Daniella Genovese of Fox Information Digital contributed to this report.
#In style #model #eye #drops #recalled , #Gossip247
5ccfae93-b75a-510e-be7c-05092dc2d0e1,fbn,Fox Enterprise,fox-business/options/product-recalls,fox-news/fox-news-newsedge/fox-news-health,fox-news/fox-news-newsedge/fox-news-business,fox-business/healthcare,article ,
ketchum
elon musk internet value
david bonderman
adobe inventory
nationwide grid
microsoft ai